This is the decade we made one of the world’s deadliest diseases preventable and curable
The commitment to research during the last two Ebola outbreaks has changed the course of this disease. Jeremy Farrar reflects on what made this possible.

For me, science is about asking not how the world is, but what it could be – asking what if.
Almost six years ago, and just a few months after I joined Wellcome as Director, a two-year-old boy died in a remote Guinean village. His death was the first in an Ebola epidemic which took more than 11,000 lives in less than two years in West Africa. It remains the world’s largest Ebola epidemic.
As 2019 draws to a close, it is in eastern provinces of the Democratic Republic of the Congo (DRC) that Ebola has been devastating communities – for 18 months, in what is now the world’s second largest outbreak.
It is not coincidence that the two largest Ebola epidemics have occurred in the last five years. This is the world we now live in, and it is this world we need to be prepared for. Each death is a tragedy, and healthcare teams face a constant threat of violence as they try to stop Ebola.
Nonetheless, something is now different – something National Geographic last week hailed among the greatest scientific achievements of the decade.
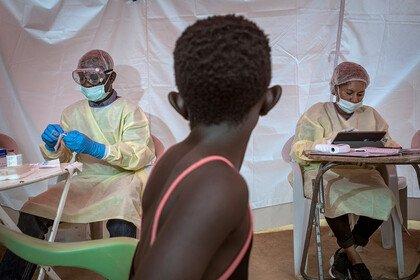
Ebola is now a disease which can be diagnosed, prevented and cured.
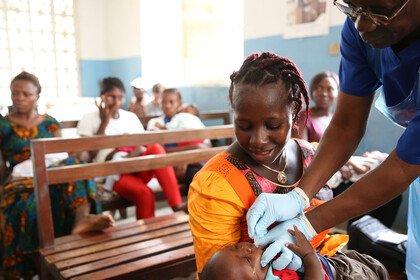
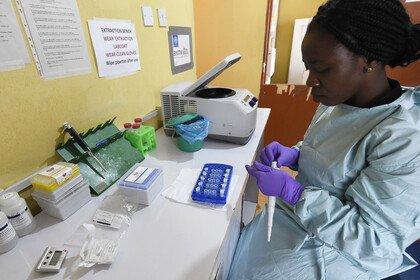
The toll in DRC is more than 2,200 deaths and continues to rise. But, since last August, more than 250,000 people in DRC have been protected by Ebola vaccination and, with development of diagnostics and treatments, there are over 1,000 Ebola survivors.
These advances have not, in fact, taken 10 years, but just a little over five. They are achievements made because in 2014 we dared to ask ‘what if’. What if we could make sure research was at the heart of every epidemic response?
There were many who doubted it would be possible.
Epidemics are among our greatest health threats, and they strike quickly. How could vaccine or treatment studies be contemplated, when they take years and require complex ethical and regulatory systems quite unsuited to the speed needed in outbreak responses? And, though a threat to us all wherever we live, epidemics often strike first in regions with the most fragile of health systems.
Despite these considerable challenges, there were researchers, governments, the World Health Organzation (WHO), aid agencies, charities, industry and funders, including Wellcome, willing to come together.
It is thanks to collaborations that in November the European Medicines Agency (EMA) approved licensure of the Merck Ebola Vaccine (Ervebo); that, earlier this year, trials of four potential treatments have identified that two significantly increase the chances of living after being infected with Ebola; and that responders now have detailed information to help them to understand and navigate the complex local contexts in which they are working.
And finally, in Rwanda, Uganda and the DRC trials of a second, complementary, preventative vaccine, from Johnson & Johnson, are underway.

Photographs taken at the beginning of December, capturing hopeful scenes of health leaders from DRC and Rwanda leading a community celebration to launch trials for a second vaccine, go a long way to encapsulating the remarkable progress.
But front of mind too is the experience of almost a year ago, when I joined Dr Tedros, Director-General of the WHO, in a visit to the affected communities in DRC. I hope to return to the region soon.
The security situation in this complex region was then – and remains – febrile. Healthcare teams are, again, under attack and the ongoing Ebola response, which was looking to have stabilised, is again under threat. There is no end to this Ebola epidemic if the violence continues. Healthcare workers must not be a target.
Our thoughts stay with all those brave and dedicated teams putting their lives at risk to protect others.
We must ensure the benefits of this research, the diagnostic tests, the vaccines, the life-saving drugs and the social science work, are available, accessible and affordable to everyone who needs them independent of their ability to pay.
And we must bring the same commitment to conducting research in every epidemic.
Only by doing so can we bring hope and turn these terrifying deadly diseases into something we can prevent, treat and no longer fear.
It is right too, however, that we reflect on what has been achieved, remember that progress is possible, that we can come together to make the world a better place.
Overview of Wellcome’s Ebola support
Since 2014 Wellcome has provided more than £41m to ensure, in partnership with others, that the world can respond to Ebola. This includes funding for development of vaccines, treatments, diagnostics and social science research.
Through partnership we expand our remit and reach. Recent funding was through the Joint Initiative on Research for Epidemic Preparedness and Response, a partnership with the UK Department for International Development (DFID).
We were also one of the founding partners and provided £80m for the Coalition for Epidemic Preparedness Innovation (CEPI), a global partnership to support development of vaccines to prevent epidemics.
- September 2014: Wellcome, in partnership with Guinea, WHO, Norway and MSF, funds a group to evaluate an Ebola vaccine produced by Merck, rVSV-ZEBOV, during the West Africa outbreak. This research demonstrates that the vaccine is safe and effective. The vaccine becomes an important tool for future Ebola responses.
- May 2016: The WHO Blueprint for R&D is established with support from Wellcome and DFID. A global strategy and preparedness group is also formed to allow for the rapid activation of research before and during epidemics. Its aim is to fast-track the availability of effective tests, vaccines, medicines and social science that can be used to save lives and avert large scale crisis.
- January 2017: The Coalition for Epidemic Preparedness Innovation (CEPI) launches, in response to calls for a new system for developing vaccines to prevent epidemics – one based on partnerships across sectors and countries. Wellcome is one of the founders.
- August 2017: Wellcome funds the Center for Infectious Disease Research and Policy to develop a research and development blueprint for Ebola and Marburg vaccines. This is intended to help guide and accelerate global efforts to develop vaccines against these diseases.
- May 2018: Wellcome awards £2 million to the WHO to support vaccine research response to the Ebola outbreak in north-western DRC. The outbreak is quickly brought under control. A team of social scientists are awarded funding to produce operational briefs setting out local, social and cultural contexts for both Ebola outbreaks. These helped shape communication with affected communities.
- March 2019: Wellcome awards £2 million to the WHO to support vaccine research response to the Ebola outbreak in eastern DRC. These funds help provide the rVSV-ZEBOV vaccine to people who have been exposed to Ebola, to their contacts, and to frontline workers who are at risk of catching the disease.
- August 2019: CEPI confirms funding to the Uganda Virus Research Institute for their ZEBOVAC J&J Ebola vaccine trial. The trial aims to enroll 800 people, focusing on healthcare workers who would be on the frontline of any Ebola response.
- October 2019: Wellcome awards £130,000 to a UNICEF and Global Outbreak Alert and Response Network (GOARN)-led partnership delivering essential social science research to support the response in the complex outbreak region in DRC.
- November 2019: Wellcome awards £2.1m funding for trials of the Johnson & Johnson vaccine in Rwanda. The vaccine is being given to over 200,000 individuals including at-risk frontline workers, such as healthcare workers and border officials, and healthy volunteers living in western districts near the border with DRC. Research will also look at how safe the vaccine is for pregnant women and young children.
- 2020: Wellcome will award funding to the Johnson & Johnson vaccine trail in DRC, which will be available to healthy volunteers who are at risk of Ebola but are outside the area currently affected by the outbreak.